Class Mammalia
Order Lagomorpha
Family Leporidae
Sylvilagus sp.—Cottontails // Sylvilagus audubonii—Desert Cottontail—Cottontails // Sylvilagus bachmani—Brush Rabbit // Sylvilagus cunicularius—Mexican Cottontail // Sylvilagus floridanus—Eastern Cottontail // Sylvilagus hibbardi—Hibbard's Cottontail // Sylvilagus nuttallii—Mountain Cottontail // Sylvilagus robustus—Davis Mountains Cottontail
The taxonomic status of Sylvilagus in the region is unsettled. An early period of naming populations in different mountainous regions was followed by consolidation to a smaller number. For a considerable length of time, three species were recognized in New Mexico (Findley et al. 1975): S. audubonii (Desert Cottontail) in the lowlands throughout the state, S. nuttallii (Nuttall's or Mountain Cottontail) in the northern mountains, and S. floridanus (Eastern Cottontail) in the southern mountains and in the far eastern grasslands.
The subspecies of S. floridanus from the Guadalupe Mountains of Texas, and thus presumably also in the Guadalupe Mountains of New Mexico, were later raised to species level (S. robustus Frey 2004), and with other populations occurring in the Davis and Chisos mountains of Texas (Schmidly 2004). However, a recent study (Nalls, et al. 2012) strongly suggested that S. robustus should be retained within S. floridanus, differences between the reputed species being more characteristic of intraspecific populations than of separate species. Nalls et al. noted that the primary difference is the larger size of the montane form. Olcott and Barry (2000) found that body size of S. floridanus varied positively in the Southwest and Mexico with high altitudes, wetter and cooler summer environments, and low variability in winter precipitation, conditions met in the Guadalupe, Davis, and Chisos mountains. For the purposes of the present work, "robustus" will be considered as a subspecies of S. floridanus.
In 1998, Ruedas resurrected two species: S. cognatus (Manzano Mountain Cottontail) and S. holzneri (Holzner's Cottontail). Both had been considered to be subspecies of S. floridanus. Frey (2004) gives the distribution of S. cognatus as occurring in the highlands of the central portion of New Mexico: Mount Taylor, Datil, Magdalena, San Mateo, Sandia, Manzano, Capitan, and Sacramento mountains. Additionally, she notes occurrences outside of the highland area in San Miguel County. The limited sample available to me at present suggests that fossil specimens would be identified as S. floridanus when utilizing the three-species concept.
The range of S. holzneri is less well defined. It occurs in mountainous terrain in the southwestern portion of New Mexico north to the Mogollon Plateau; the status of those within the Plateau area is unknown (Frey 2004).
In the regional literature, identifications of cottontails from the fossil record have been in terms of the traditional three (plus one identification of S. robustus by Dalquest and Stangl 1984b), with no recognition of differences among the three groups traditionally identified as S. floridanus. At present, post-cranial material is unidentifiable to species. Isolated third lower premolars and partial dentaries with and without teeth are the specimens most often available for identification. Much of the identification is based on characters of p3. Typical differences are shown in Fig. 1, but there is a fair amount of intraspecific variation and a degree of overlap between species.
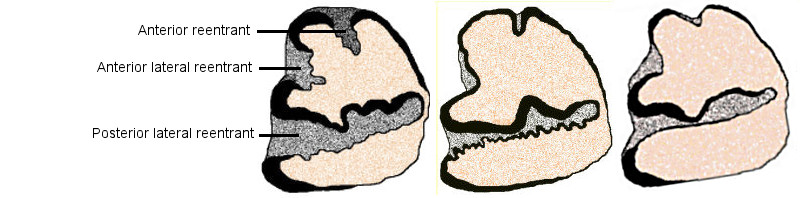
Fig. 1. Drawings of more or less typical lower third premolars of (left to right) S. audubonii, S. floridanus (S. cognatus/S. robustus) and S. nuttallii, showing differences in configuration. Dentine orange, enamel black, and cementum gray.
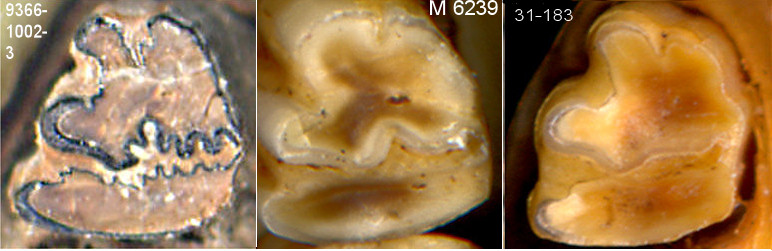
Fig. 2. Images of the lower third premolars of same three species as in Fig. 1 (different individuals); the image of the S. floridanus specimen is a right premolar reversed for comparison to the two left premolars.
Extensive study of fossil Sylvilagus during October and early November of 2010 has clarified identifications and the identification process. All specimens used in that study have at least six characters measured or coded: alveolar length of dentary toothrow; dentary depth at p/4; depth of anterior reentrant of p/3 (taken with an eyepiece micrometer and measured as the distance from the posteriormost enamel of p/3 to the most posteriormost enamel of the anterior reentrant; the figures in the basic statistics and used in the analyses are a multiple of three of the actual distance); degree of crinkling of the anterior enamel of the posterior lateral reentrant of p/3 (coded as "1" no strong indentation, as "2" if no more than 1 loop or strong indentation, and as "3" if there are two or more strong loops); crinkling of the posterior enamel of the posterior lateral reentrant (coded as "1" if none, and "3" if crinkling is present); and the same coding for the anterior enamel of the talonid of post-p/3 teeth. In addition, most modern specimens and several fossil specimens supplied three additional measurements: minimum width of the basioccipital, maximum width of the basioccipital, and alveolar length of the upper toothrow. In preliminary sorting, all available measurements were utilized. Modern identified specimens were used as a starting point, with fossil specimens added to the samples as various combinations of techniques identified them with reasonable likelihood.
Using combinations of modern and fossil specimens that had the first six measurements available (and with some having an additional three), 43 S. nuttallii, 18 S. floridanus, and 34 S. audubonii were used for characterization of the species.
Sylvilagus nuttallii is separable from the other two species using the first six characters by principal components analyses (PCA) and by clustering techniques. The non-crinkling of the anterior enamel of the posterior lateral reentrant and of post-p3 character state (lack of crinkling) are the major factors discriminating S. nuttallii. Sites having S. nuttallii by these criteria are Harris' Pocket, Dry Cave [UTEP Loc. 6]; Animal Fair, Dry Cave [UTEP Loc. 22] (full-glacial and near-glacial levels); Camel Room, Dry Cave [UTEP Loc. 25]; Human Corridor, Dry Cave [UTEP Loc. 31]; Howell's Ridge Cave [UTEP Loc. 32]; Isleta Cave No. 2 [UTEP Loc. 46]; Dark Canyon Cave [UTEP Loc. 75]; Conkling Cavern [UTEP Loc. 90]; Algerita Blossom Cave [UTEP Loc. 119]; and U-Bar Cave [UTEP Loc. 5689] (mid and late Wisconsin).
Sylvilagus audubonii and S. floridanus overlap in cluster analysis and principal components analysis, but are separable by discriminant analysis. Sites having S. audubonii using these criteria are Balcony Room, Dry Cave [UTEP Loc. 12]; Khulo Site [UTEP Loc. 21]; Isleta Cave No. 1 [UTEP Loc. 41]; and Isleta Cave No. 2 [UTEP Loc. 46]; U-Bar Cave [UTEP Loc. 5689], late Wisconsin; and Conkling Cavern [UTEP Loc. 90]. Sites having S. floridanus by these criteria are Room of the Vanishing Floor, Dry Cave [UTEP Loc. 26]; Above Room of the Vanishing Floor, Dry Cave [UTEP Loc. 27]; Isleta Cave No. 1 [UTEP Loc. 41]; and Isleta Cave No. 2 [UTEP Loc. 46].
For specimens consisting of p/3, S. nuttallii is clearly separable from S. floridanus and S. audubonii by principal components analysis and by clustering; the latter two species are not discriminated by either of these techniques nor by discriminant analysis—the degree of overlap is simply too great. However, extremes usually can be identified, with S. audubonii characteristically having heavy crinkling of both the anterior and posterior enamel of the posterior lateral reentrant and with the projection between the anterior reentrant and the anterior lateral reentrant relatively narrow and hooked laterally (as seen in Fig. 2). Sylvilagus floridanus normally lacks this hooked configuration and the anterior enamel of the posterior lateral reentrant tends to be less crinkled—however, there is considerable overlap in this and other measurements between the two species.
The multivariate analyses pictured here utilize the p/3 characters plus the alveolar length of the dentary toothrow and the depth of the dentary at p4.
Other characters of use are bulla size (Fig. 3) when bullae are available: large in S. audubonii, intermediate in S. nuttallii, and small in S. floridanus.
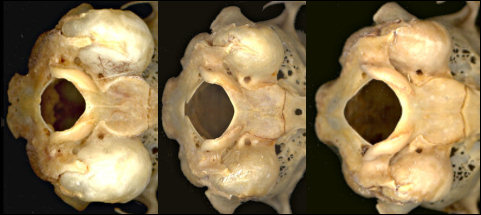
Fig. 3. Posterior ventral views of (left to right) S. audubonii, S. nuttallii, and S. floridanus cognatus, showing differences in relative bulla sizes. Also note the constriction of the basioccipital in S. audubonii.
Sylvilagus audubonii: The walls of the interpterygoid fossa are slightly ridged, unlike in the other species. The depth of the dentary generally is greater than in the other two (Findley et al. 1975), although Neusius and Flint (1985) did not find its utility as great as hoped. Specimens of modern and fossil Sylvilagus used here are plotted in Fig. 4. The lower third premolar almost always is strongly crenulated; the anterior reentrant usually is deep and the external lobe thus formed normally is narrow and bent laterally anteriorly. The auditory bullae are large and the constriction of the basioccipital in relation to its greatest width is extreme (Fig. 3).
Sylvilagus floridanus: The dentary depth is relatively slight and the auditory bullae are small. However, the dentary depth will not separate this taxon from S. nuttallii and the strength of the tooth crenulations overlaps the conditions in S. audubonii, though averaging less.
The S. floridanus modern sample from the Southwest is small and needs to be augmented. A sample of modern specimens from Kansas strongly skew the S. floridanus sample by their generally larger size and are not used here. The small sample available of western S. floridanus (presumably equals S. holzneri) are quite different from the small modern sample of S. floridanus from the southeastern and eastern parts of New Mexico. A single high-elevation specimen from the Manzano Mountains (S. f. cognatus) fits with the western group and is especially close to a specimen from 1 mile north of Mimbres, in Grant County.
Sylvilagus nuttallii: This is the easiest of the cottontails to identify. Crenulations are lacking or of very low grade in all the cheekteeth; this usually will separate the taxon from the others. The dentary depth normally separates this species from the deeper dentary condition of S. audubonii.
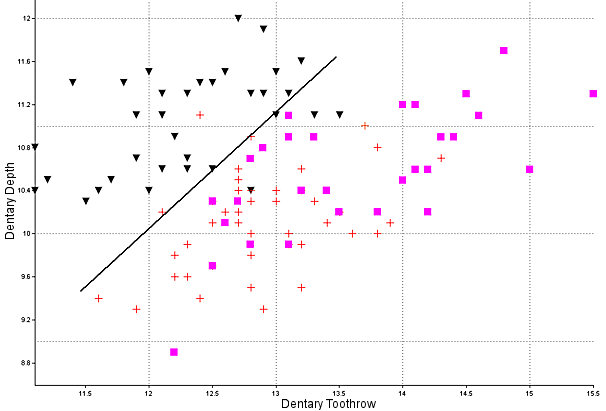
Fig. 4. Alveolar length of the dentary toothrow of cottontails (modern and fossil) plotted against the depth of the dentary at p4. Triangles, S. audubonii; crosses, S. nuttallii; squares, S. floridanus. All the S. floridanus specimens with a toothrow length of 14 mm or more are from Kansas. The line shows the approximate best separation of S. audubonii from the other two species.
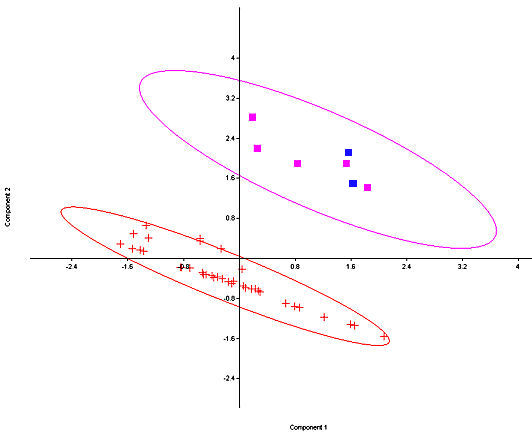
Fig. 5. Principal Components plot of fossil and modern S. nuttallii (crosses) and S. floridanus (squares, with the two fossil specimens from Isleta Cave No. 1 in blue) with 95% ellipses. Only Southwestern specimens of S. floridanus are utilized. Components 1 and 2 account for 65% of the variance.
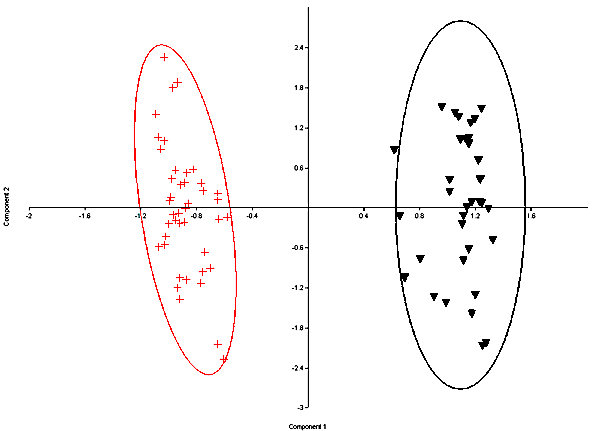
Fig. 6. Principal Components plot of fossil S. nuttallii (crosses) and S. audubonii (triangles)with 95% ellipses. Both modern and fossil specimens are represented. Components 1 represents 76% and Component 2 represents 12% of the variance.
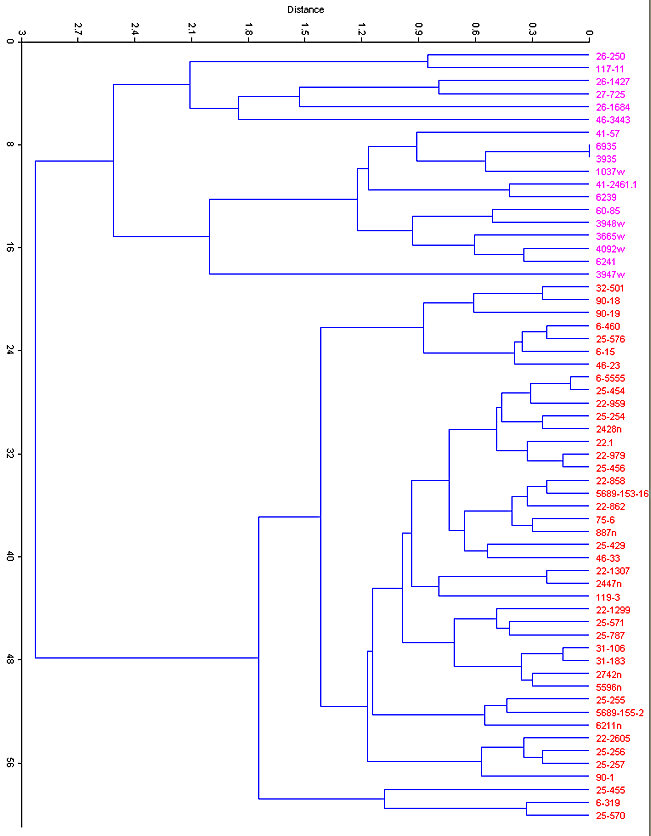
Fig. 7. Cluster analysis (Euclidean distance) of modern and fossil specimens of S. floridanus (magenta) and S. nuttallii (red) based on six variables of the lower jaw.
Basic statistics are available for modern and fossil samples.
Statistics software used for this page is PAST (Hammer, et al. 2001).
Literature. Dalquest and Stangl 1984b; Findley et al. 1975; Frey 2004; Hammer et al. 2001; Nalls, Ammerman, and Dowler 2012; Neusius and Flint 1985; Olcott and Barry 2000; Ruedas 1998; Schmidly 2004.
Last Update: 10 Mar 2013