clinical project
Clinical Project
The El Paso-Juarez Borderplex, and the University of Texas at El Paso
The El Paso/Juarez Borderplex presents a unique and critical ecosystem for studying salient aspects of Mexican American health, based on the: large diversity of Mexican American immigrant generations; wide variations in residential, social, and institutional contexts in which individuals live; presence of some of the poorest rural “colonias” in the US and Mexico along with urban neighborhoods with high poverty as well as urban and suburban communities with high concentrations of Mexicans and Mexican Americans with high incomes; and complex commuting dynamics of persons from both sides of the border indicative of significant shared cultural, familial, and economic indices. The UTEP is a preeminent institution focusing on health and biomedical issues affecting the Mexican origin population in the Borderplex region. The UTEP BBRC, supported by the National Institutes of Health (NIH) Research Centers in Minority Institutions (RCMI) program since 1992, is the cornerstone for biomedical studies and provides state-of-the-art analytical and experimental support. The demographic characteristics of the El Paso/Juarez Borderplex, the advanced laboratory and analytical capabilities of the BBRC, and the outstanding working interactions between medical and academic partners, provide robust competitive advantages for developing basic, behavioral, and clinical research programs at UTEP.
Cancer is the second leading cause of death in the United States and “health disparities” contribute to cancer initiation, development, incidence, prevalence, and severity. The National Cancer Institute (NCI) defines cancer health disparities as “adverse differences in cancer incidence, prevalence, death, survivorship, and burden of cancer or related health conditions that exist among specific population groups in the United States (1).” A key population characteristic is defined by race and ethnicity, and cancer exacts a marked detrimental effect on the Hispanic demographic. In fact, and as first reported in 2012, cancer is the leading cause of death for Hispanics in the US (2). In addition, and as described below, Hispanics suffer from marked cancer health-related disparities, which can be divided into three main categories.
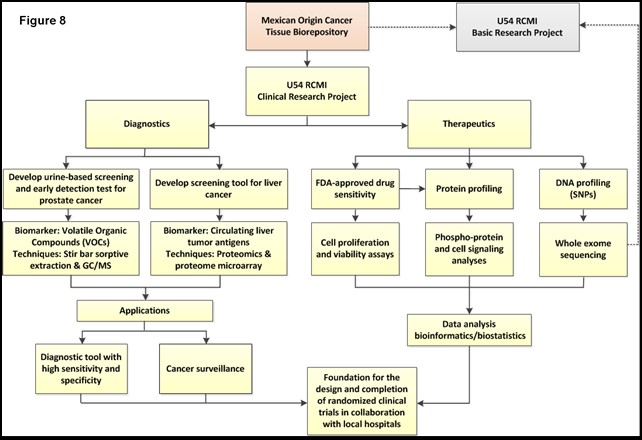
Specific Aim 1: Establish the first comprehensive Mexican Origin Cancer Tissue Biorepository in the BBRC with tissues procured from Mexican origin patients.
Specific Aim 2: Develop customized treatment strategies for relapse cancer patients in a predominately Mexican origin Hispanic population by integrating genetic, molecular, and drug sensitivity signatures as platforms to deliver targeted therapeutic approaches.
Specific Aim 3: Develop early detection signatures for prostate and Liver Cancers.
Impact: The strategic integration of cancer tissue samples; genetic, molecular and physiological markers; therapeutic interventions; and diagnostic tool development, provides a novel and innovative strategy for investigating the complex conditions that underpin Hispanic cancer health disparities. The current research findings will exert a sustained and powerful influence on the field by providing novel insight and direction for reducing the deleterious effects of cancer in Hispanics of Mexican origin, and will establish The UTEP BBRC and the El Paso/Juarez Borderplex as critical drivers of Hispanic cancer health disparities research.
Investigative Team
Jianying Zhang, M.D., Ph.D. (Principal Investigator):
Dr. Zhang is a Professor in the UTEP Department of Biological Sciences. His research career has focused on the identification, characterization and evaluation of tumor-associated antigens as biomarkers in cancer detection, and he has expertise in the molecular, histopathological, immunological, and proteomic approaches proposed in this application for cancer biomarker research. Dr. Zhang will also serve as lead scientist on the liver cancer subproject under the Diagnostics arm of this project.
Wen-Yee Lee, Ph.D. (Co-Investigator):
Dr. Lee is an Associate Professor in the UTEP Department of Chemistry and Biochemistry and will serve as the lead scientist on the prostate cancer subproject of the Diagnostics arm of this proposal, and will hold primary responsibility for directing and managing all aspects of project implementation. She will work on all chemical analyses and will work with the Research Associate (RA) on the analysis of volatile organic compounds (VOCs) in urine, organization of experiments, interpretation of data, and publication of results. She will meet regularly with key personnel to track and facilitate progress and discuss the results of the research.
Elisa Robles-Escajeda, Ph.D. (Contributor):
Dr. Robles-Escajeda is a Postdoctoral Researcher in the UTEP Department of Biological Sciences, with significant biomedical research experience. Dr. Robles-Escajeda is also a Mexican trained physician. Her research has focused on the analysis of cancer tissue and the assessment
of the drug sensitivity models, protein expression and cell signaling in cancer cells. Dr. Robles-Escajeda will coordinate efforts with local hospitals and will execute the development of a comprehensive Biorepository and the collection of clinical data.
Armando Estrada, Ph.D. (Contributor):
Dr. Estrada is a Postdoctoral Researcher in the UTEP Department of Biological Sciences, with expertise in molecular biology and immunology. He has been characterizing signaling pathways in T cells responsible for T-cell derived pathologies such as leukemia and characterizing protein signaling pathways that may serve as therapeutic targets to treat relapse tumors. Furthermore, he also performs FDA approved drug sensitivity assays on relapse cancer cells to identity drug candidates that may be utilized as second line therapy for relapse cancer patients.
- National Cancer Institute. Cancer Disparities. 2018; Available from: this source
- American Cancer Society. Cancer Facts and Figures for Hipsanics/ Latinos 2015-2017. 2015.




