Current Projects
ln-doped Tin Pyrophosphates as Efficient Intermediate-Temperature Fuel-Cell Electrolytes
PI:Dr. Cristian E. Botez (Physics)
Students:Heber Martinez (MASE Ph.D. Interdisciplinary Program)
 Project Description: Novel functional materials that can operate as fuel-cell electrolytes at intermediate temperatures (within the 150⁰C - 500⁰C range) are of particular importance for large scale clean-energy applications (e.g. in the automotive industry), as well as for more specific applications based on the portability and low noise of these devices. Good candidate materials have to exhibit a large proton conductivity at intermediate temperatures, and, among them, In-doped tin pyrophosphates (InxSn1-xP2O7) present an interesting case. Their enhanced proton conductivity is not due to a polymorphic structural transition (as for their solid-acid counterparts); similar structures have been reported for all doping levels and operating temperatures. Yet, the In0.1Sn0.9P2O7 (x=0.1) compound has the highest proton conductivity within the series, and understanding the subtle crystallographic modifications that occur when exactly 10% of the Sn atoms are replaced by In is necessary in order to reveal the microscopic details that enhance the proton transport. Eventually, these findings could be used to tune the proton conductivity via chemical manipulation, with the eventual goal of synthesizing more efficient intermediate temperature fuel cell electrolytes.
Project Description: Novel functional materials that can operate as fuel-cell electrolytes at intermediate temperatures (within the 150⁰C - 500⁰C range) are of particular importance for large scale clean-energy applications (e.g. in the automotive industry), as well as for more specific applications based on the portability and low noise of these devices. Good candidate materials have to exhibit a large proton conductivity at intermediate temperatures, and, among them, In-doped tin pyrophosphates (InxSn1-xP2O7) present an interesting case. Their enhanced proton conductivity is not due to a polymorphic structural transition (as for their solid-acid counterparts); similar structures have been reported for all doping levels and operating temperatures. Yet, the In0.1Sn0.9P2O7 (x=0.1) compound has the highest proton conductivity within the series, and understanding the subtle crystallographic modifications that occur when exactly 10% of the Sn atoms are replaced by In is necessary in order to reveal the microscopic details that enhance the proton transport. Eventually, these findings could be used to tune the proton conductivity via chemical manipulation, with the eventual goal of synthesizing more efficient intermediate temperature fuel cell electrolytes.
Need for x-ray methods / use of XRSLab: High-resolution powder diffraction is an ideal tool for the above-mentioned structural studies because of its ability to reveal subtle crystallographic modifications with sub-Ångstrom resolution. In addition, the controlled-sample-environment capabilities afforded by the XRK reactor allows us to investigate the crystal structures (and structure modifications) of the title materials at different temperatures and under different gas atmospheres (flow, pressure).
High-Temperature Phase Transitions in CsH2PO4 and RbH2PO4/Silica composites
PI:Dr. Cristian E. Botez (Physics)
Students:Israel Martinez (MASE Ph.D. Interdisciplinary Program), Adan Anchondo (Engineering, Masters Interdisciplinary Program), Victor Gonzalez (Biology, Undergraduate Interdisciplinary Program), Jacob Fong (Physics, Undergraduate Interdisciplinary Program).
 Project Description: The sudden increase in the proton conductivity of CsH2PO4 and RbH2PO4 upon heating above a temperature threshold renders these compounds attractive for intermediate temperature fuel cell applications. Yet, the instability of the high-temperature superprotonic phase that occurs via dehydration under ambient pressure and humidity is a major roadblock to the application of the title materials as fuel cell electrolytes on a large scale. Combining these compound dispersed silica has been proposed as a possible way to address this issue. In this project we are investigating the effect of mixing solid acids CsH2PO4 and RbH2PO4 with dispersed silica (SiO2) on the properties of their superprotonic phases. The approach to achieve this goal will include: 1) determining the relationship between the composition and microstructure of the (1-x)CsH2PO4/xSiO2 and (1-x)RbH2PO4/SiO2 composites and the proton conductivity of their high temperature phases. 2) determining the thermal behavior of the solid-acid component upon heating under ambient and high-pressure, as a function of the silica content and average grain size.
Project Description: The sudden increase in the proton conductivity of CsH2PO4 and RbH2PO4 upon heating above a temperature threshold renders these compounds attractive for intermediate temperature fuel cell applications. Yet, the instability of the high-temperature superprotonic phase that occurs via dehydration under ambient pressure and humidity is a major roadblock to the application of the title materials as fuel cell electrolytes on a large scale. Combining these compound dispersed silica has been proposed as a possible way to address this issue. In this project we are investigating the effect of mixing solid acids CsH2PO4 and RbH2PO4 with dispersed silica (SiO2) on the properties of their superprotonic phases. The approach to achieve this goal will include: 1) determining the relationship between the composition and microstructure of the (1-x)CsH2PO4/xSiO2 and (1-x)RbH2PO4/SiO2 composites and the proton conductivity of their high temperature phases. 2) determining the thermal behavior of the solid-acid component upon heating under ambient and high-pressure, as a function of the silica content and average grain size.
Need for x-ray methods / use of XRSLab: The high - resolution capabilities of the Empyrean XRD system, coupled with the controlled-sample-enviroment of XRK reactor and ultrafast PixCell detector will allows us to collect the time- and temperature-resolved data. Analysis (LeBail, Pawley and Rietveld refinements) will be performed using the High-Score software, also available in the XRSLab.
Lanthanide-doped Cu and Al delafossite oxides as transparent conductive materials
PI:Dr. Cristian E. Botez (Physics)
Postdoctoral Researcher:Dr. Alan Goos
Student:Golam Mustafa (MASE Ph.D. Interdisciplinar Program)
 Project Description: Since their discovery in 1997 as p-type transparent conductors, CuAlO2 films with delafossite structure have been the subject of many studies. More recently, several other ABO2 delafossite materials have been synthesized and investigated. To date, ABO2 compounds (A = Ag, Cu; and B = B, Al, Ga, In, Fe, Cr, Sc, Y) have been found to exhibit high p-type conductivity and high optical transparency, which renders them particularly useful for a wide variety of applications, from displays to solar cells. Doping these compounds with trivalent lanthanide metals may impart novel magnetic, optical, and conductive properties. This project uses hydrothermal and solid-state synthesis techniques to synthesize CuAlO2 and explore the possibility of Sm and Y doping at levels that would preserve the delafossite structure but impart new macroscopic properties.
Project Description: Since their discovery in 1997 as p-type transparent conductors, CuAlO2 films with delafossite structure have been the subject of many studies. More recently, several other ABO2 delafossite materials have been synthesized and investigated. To date, ABO2 compounds (A = Ag, Cu; and B = B, Al, Ga, In, Fe, Cr, Sc, Y) have been found to exhibit high p-type conductivity and high optical transparency, which renders them particularly useful for a wide variety of applications, from displays to solar cells. Doping these compounds with trivalent lanthanide metals may impart novel magnetic, optical, and conductive properties. This project uses hydrothermal and solid-state synthesis techniques to synthesize CuAlO2 and explore the possibility of Sm and Y doping at levels that would preserve the delafossite structure but impart new macroscopic properties.
Need for x-ray methods / use of XRSLab: Clearly, x-ray methods are the most accurate in terms investigating the crystal structures of the title materials and confirming the persistence of the delafossite structure upon doping. Single crystal and powder x-ray diffraction data analysis will reveal the microscopic aspects of the dopant incorporation, as well as possible minute crystallographic distortions responsible for the sought macroscopic property modifications. Moreover, temperature-induced phase transitions and behaviors under non-ambient gas atmospheres will be investigated.
Anion-directed Supramolecular Aromatic Networks of Silver (I) N-(4-pyridyl) Benzamide and Their Band Gap Tunability Properties
PI:Dr. Juan Noveron (Chemistry)
Students: Isaac Torres (BS in Chemistry), Noemi Dominguez (MASE Ph.D.)
 Project Description: We are working on a project regarding supramolecular materials. Supramolecular materials are based on non-covalent bonds, involving weak and reversible molecular interactions, hydrogen bonding, metal-ion coordination, dipole-dipole association, Van der Waals and hydrophobic forces, among others. Additionally, these interactions allow the buildup process (building blocks) of systems with well-ordered and functional structures at nano and micro-scale as well as at macro-scale. These materials can be used in a wide range of applications such as medicine, electronic and optoelectronic devices, liquid crystalline displays (LCDs), organic light-emitting diodes, organic photovoltaic cells, among others.
Project Description: We are working on a project regarding supramolecular materials. Supramolecular materials are based on non-covalent bonds, involving weak and reversible molecular interactions, hydrogen bonding, metal-ion coordination, dipole-dipole association, Van der Waals and hydrophobic forces, among others. Additionally, these interactions allow the buildup process (building blocks) of systems with well-ordered and functional structures at nano and micro-scale as well as at macro-scale. These materials can be used in a wide range of applications such as medicine, electronic and optoelectronic devices, liquid crystalline displays (LCDs), organic light-emitting diodes, organic photovoltaic cells, among others.
Need for x-ray methods / use of XRSLab: We need to obtain the x-ray patterns of three different supramolecular compounds.The x-ray diffraction powder technique in this project is crucial. We want to compare and match these x-ray powder patterns with the simulated x-ray powder diffraction patterns from single crystal data.
Self-assembly of Metal-Organic Solar Cells
PI:Dr. Juan Noveron (Chemistry)
Students: Rudy Acosta (BS in Chemistry)
 Project Description: The goal of this project is to develop novel supramolecular fullerene nanostructures with unusual designs and generate a library of nanostructure-function relationships with regards to multiple-heterojunctions and multiple-exciton-generation processes in organic solar cells. According to the Shockley−Queisser Limit, solar cells with a typical single heterojunction have a theoretical power conversion efficiency of 37%; however, this limit can be extended to a theoretical 83% efficiency via multiple heterojuctions that have well-orchestrated photoelectric pathways that cover a wide-spectrum of radiation. Similarly, this limit may double when the system promotes multiple exciton generation (MEG) processes in which a single-photon can generate multiple electron-hole pairs. The MEG mechanism has only been sparsely investigated and it involves a single-photon that generates a special exciton (electron-hole pair) that undergoes exciton fission and splits into triplet excitons, which if long-lived and diffuse to the right nanostructure, turns into electrical potential and generates a current. MEG has been demonstrated with inorganic crystals, but there is an interest in developing new organic materials that exhibit MEG because of their relative low-cost and flexibility. Multi-junctions and MEG represent the next leap in photovoltaic research, but require well-organized nanostructures where molecules with the right photo- and electro-chemical properties interact. Although MEG processes have not been demonstrated in working solar PV cells, it has recently been confirmed in bis(tetracene) molecules73 and C60-pentacene bilayers, indicating that MEG may also operate in other organic semiconductors. In particular, the C60-pentacene bilayer study revealed that the precise nanoscale positioning of components that have suitable photo-electrochemical properties are prerequisites for the formation of long-lived triplet excitons (200ns), which are essential for MEG processes to occur. Similar long-lived excitons have been observed in fullerene compounds. In this context, we propose to investigate new methods that lead to supramolecular fullerene structures and study their electron-transfer processes as a function of the nanoscale architectures. In this project, we will develop a library of new supramolecular fullerene nanostructures using pre-configured self-assembly and supramolecular post-modifications in the crystalline solid-state and study their physical and chemical properties with regards to photoelectron-transfer reactions.
Project Description: The goal of this project is to develop novel supramolecular fullerene nanostructures with unusual designs and generate a library of nanostructure-function relationships with regards to multiple-heterojunctions and multiple-exciton-generation processes in organic solar cells. According to the Shockley−Queisser Limit, solar cells with a typical single heterojunction have a theoretical power conversion efficiency of 37%; however, this limit can be extended to a theoretical 83% efficiency via multiple heterojuctions that have well-orchestrated photoelectric pathways that cover a wide-spectrum of radiation. Similarly, this limit may double when the system promotes multiple exciton generation (MEG) processes in which a single-photon can generate multiple electron-hole pairs. The MEG mechanism has only been sparsely investigated and it involves a single-photon that generates a special exciton (electron-hole pair) that undergoes exciton fission and splits into triplet excitons, which if long-lived and diffuse to the right nanostructure, turns into electrical potential and generates a current. MEG has been demonstrated with inorganic crystals, but there is an interest in developing new organic materials that exhibit MEG because of their relative low-cost and flexibility. Multi-junctions and MEG represent the next leap in photovoltaic research, but require well-organized nanostructures where molecules with the right photo- and electro-chemical properties interact. Although MEG processes have not been demonstrated in working solar PV cells, it has recently been confirmed in bis(tetracene) molecules73 and C60-pentacene bilayers, indicating that MEG may also operate in other organic semiconductors. In particular, the C60-pentacene bilayer study revealed that the precise nanoscale positioning of components that have suitable photo-electrochemical properties are prerequisites for the formation of long-lived triplet excitons (200ns), which are essential for MEG processes to occur. Similar long-lived excitons have been observed in fullerene compounds. In this context, we propose to investigate new methods that lead to supramolecular fullerene structures and study their electron-transfer processes as a function of the nanoscale architectures. In this project, we will develop a library of new supramolecular fullerene nanostructures using pre-configured self-assembly and supramolecular post-modifications in the crystalline solid-state and study their physical and chemical properties with regards to photoelectron-transfer reactions.
Need for x-ray methods / use of XRSLab: The state of the art high-resolution single crystal diffraction is an ideal tool for sample characterisation for structural studies.
In-situ crystal structure modifications in novel nanocomposites capacitor materials
PI:Dr. Yirong Lin (Mechanical Engineering)
Students: Mohammad Shuvo (ESE Ph.D. Program)
 Project Description: To date, most of the commercially available capacitors for high temperature and pressure applications are ceramic based; however, they are fragile, relatively low energy density, and of high cost. One of the approaches to alleviate these issues is to develop nanowire reinforced polymer matrix film capacitors. The reinforced nanowire is the key to achieve high dielectric constant and high energy density for the capacitor, mainly because of the dipole movement in its perovskite structure. The dipole mobility can be affected significantly by external temperature and pressure. Therefore, it is essential to investigate the temperature and pressure influence on the dielectric characteristic that stems from their crystal structures. Testing results coupled with energy density measurements will be used to fully understand the fundamental relationships of (temperature/pressure)-structure-property, guiding the design for nanocomposites towards optimized performance.
Project Description: To date, most of the commercially available capacitors for high temperature and pressure applications are ceramic based; however, they are fragile, relatively low energy density, and of high cost. One of the approaches to alleviate these issues is to develop nanowire reinforced polymer matrix film capacitors. The reinforced nanowire is the key to achieve high dielectric constant and high energy density for the capacitor, mainly because of the dipole movement in its perovskite structure. The dipole mobility can be affected significantly by external temperature and pressure. Therefore, it is essential to investigate the temperature and pressure influence on the dielectric characteristic that stems from their crystal structures. Testing results coupled with energy density measurements will be used to fully understand the fundamental relationships of (temperature/pressure)-structure-property, guiding the design for nanocomposites towards optimized performance.
Need for x-ray methods / use of XRSLab: Currently, we have successfully fabricated dielectric nanowire reinforced polymer capacitors that are suitable for high temperature applications. We plan to investigate the temperature and pressure influence on crystal structures of samples with different types of piezoelectric nanowire and high temperature polymer combinations, e.g. Pb(Mg1/3Nb2/3)-PbTiO3 nanowire embedded in polyimide polymer matrix. To explore the influence of temperature and pressure on the dielectric constant and energy density of the novel nanocomposites capacitor, the in-situ crystal structures transformation under difference temperature and pressure must be understood.
Analysis of the anomalies found in bismuth ferrite at reported temperatures
PI:Dr. Cristian Botez (Physics)
Students: Karen Santillan (B.S. Cellular and Molecular Biochemistry)
 Project Description: Bismuth ferrite is an inorganic chemical compound with perovskite structure and one of the most promising multiferroic materials; this compound has several applications in the fields of magnetism, spintronics, photovoltaics, and gas-sensing properties, however previous studies have found that bismuth ferrite is very prone to show parasitic phases that tend to nucleate at grain boundaries and impurities, anomalies in this compound's dielectric constant and thermal expansion have been registered between room temperature and 900ºC at 130, 200, 280, 370, 460, 600, 670, 740, and 845ºC and while some of this anomalies can be explained by known phase transitions (845ºC normal bismuth ferrite phase transition) for the rest it has been theorized that said anomalies might be caused by parasitic phases due to impurities such as Bi2O3, bismuth-rich and bismuth-deficient regions in the compound.
Project Description: Bismuth ferrite is an inorganic chemical compound with perovskite structure and one of the most promising multiferroic materials; this compound has several applications in the fields of magnetism, spintronics, photovoltaics, and gas-sensing properties, however previous studies have found that bismuth ferrite is very prone to show parasitic phases that tend to nucleate at grain boundaries and impurities, anomalies in this compound's dielectric constant and thermal expansion have been registered between room temperature and 900ºC at 130, 200, 280, 370, 460, 600, 670, 740, and 845ºC and while some of this anomalies can be explained by known phase transitions (845ºC normal bismuth ferrite phase transition) for the rest it has been theorized that said anomalies might be caused by parasitic phases due to impurities such as Bi2O3, bismuth-rich and bismuth-deficient regions in the compound.
Need for x-ray methods / use of XRSLab: X-ray powder diffraction is the ideal method to examine the effect of impurities on the phase anomalies mentioned. Fine control of temparatures and fast collection available in the XRSLab ideal.
Chemical and Structural Characterization of Omeprazole at High Temperatures
PI:Dr. Cristian Botez (Physics)
Students: Manuel Fernando Martinez (B.S. Electrical and Computer Engineering)
 Project Description: Omeprazole is a proton pump inhibitor that reduces the amount of acid produced in the stomach and is used to treat dyspepsia (heartburn), peptic ulcer disease, gastroesophageal reflux disease. Omeprazole is distributed world-wide as both a prescription drug and as an over-the-counter one.Given its recommended storage temperature of between 15º and 30ºC, and the wide use of this medicine, a structural analysis of the drug under high temperatures and varying levels of pressure is necessary to understand the changes that it may undergo under these conditions. Even though the drug's average melting point (based on the different brands and manufacturers) is around 156.2ºC, the active ingredient may still undergo a series of structural and chemical changes that can affect its efficiency.
Project Description: Omeprazole is a proton pump inhibitor that reduces the amount of acid produced in the stomach and is used to treat dyspepsia (heartburn), peptic ulcer disease, gastroesophageal reflux disease. Omeprazole is distributed world-wide as both a prescription drug and as an over-the-counter one.Given its recommended storage temperature of between 15º and 30ºC, and the wide use of this medicine, a structural analysis of the drug under high temperatures and varying levels of pressure is necessary to understand the changes that it may undergo under these conditions. Even though the drug's average melting point (based on the different brands and manufacturers) is around 156.2ºC, the active ingredient may still undergo a series of structural and chemical changes that can affect its efficiency.
Need for x-ray methods / use of XRSLab: X-ray powder diffraction is a highly effective tool to perform the above-mentioned structural analysis of Omeprazole since it allows for a high resolution image of the crystallographic changes the sample may undergo. Furthermore, the technical capabilities of the PANalytical Empyrean Powder XRD machine allow for precise control of sample-enviroment variables such as temperatures and pressure that allow us to investigate the crystal structures and modifications of the title material.
The hunt for 'Fe=O':Synthetic models to better understand biomimetic intermediates
PI:Dr. Skye Fortier (Chemistry)
Students: Arnab K Maity (postdoc)
 Project Description: Highvalent iron (III/IV/V) oxxo species have been spectroscopically identified as active intermediates as active intermediates in the catalytic cycles of number of enzymatic systems. Heme and non-heme enzymes primarily use dioxygen from nature to oxidize their substrates and in the process they transiently from iron oxo intermediates. The synthesis of well-described model compounds can provide vital insights in to the mechanism of such enzymatic processs. We, in particular, are trying to develop such biomimetic models using a 'super-bulky' . guanidinate platform on iron. We have already publshed the synthesis and characterization of the 'super-bulky' guanidinate and are now in pursuit of its chemistry on iron. In this context, we are in process to use several 'oxo'-transfer reagents (e.g. N-methylmorpholine N-Oxide, Pyridine N-oxide, Trimethylamine N-oxide, (2, 2, 6, 6-Tetramethylpiperidin-1-yl)oxyl or TEMPO, iodosylbenzene or PhIO,etc) on 'super-bulky-iron' to generate 'super-bulky-iron-oxo'species. One such reaction with TEMPO has given us some promising results, and if we are successful, we will be able to study more about such rare 'iron-oxo' species. In broader sense that will help us and the bioinorganic community to understand the enzymatic processes.
Project Description: Highvalent iron (III/IV/V) oxxo species have been spectroscopically identified as active intermediates as active intermediates in the catalytic cycles of number of enzymatic systems. Heme and non-heme enzymes primarily use dioxygen from nature to oxidize their substrates and in the process they transiently from iron oxo intermediates. The synthesis of well-described model compounds can provide vital insights in to the mechanism of such enzymatic processs. We, in particular, are trying to develop such biomimetic models using a 'super-bulky' . guanidinate platform on iron. We have already publshed the synthesis and characterization of the 'super-bulky' guanidinate and are now in pursuit of its chemistry on iron. In this context, we are in process to use several 'oxo'-transfer reagents (e.g. N-methylmorpholine N-Oxide, Pyridine N-oxide, Trimethylamine N-oxide, (2, 2, 6, 6-Tetramethylpiperidin-1-yl)oxyl or TEMPO, iodosylbenzene or PhIO,etc) on 'super-bulky-iron' to generate 'super-bulky-iron-oxo'species. One such reaction with TEMPO has given us some promising results, and if we are successful, we will be able to study more about such rare 'iron-oxo' species. In broader sense that will help us and the bioinorganic community to understand the enzymatic processes.
Need for single crystal x-ray diffraction method: As our ‘super bulky iron’ species are paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization is ambiguous for determining the composition and connectivity/structure of the complexes. Other characterization techniques like infrared (IR), ultraviolet-visible (UVVis), electron paramagnetic resonance (EPR) are also gave us inconclusive data for the structure/connectivity of the complexes. For such paramagnetic iron complexes single crystal diffraction study is found to be most successful. In this context, we have grown some single crystals of two products from a ‘super bulky iron + TEMPO’ reaction suitable for X-ray diffraction. The crystal structure determination will conclusively tell us about the
identity of these products. This reaction yields two different paramagnetic products that crystallize as colorless and purple-plates. A request is being submitted to identify the composition of the pink and colorless crystals.
*These compounds are highly air and water sensitive and will require collection under an N2 cryostream. Liquid N2 can be provided upon request.
A Cobalt Complex Supported by a Highly Encumbering Ligand
PI:Dr. Skye Fortier (Chemistry)
 Project Description: We recently reported the synthesis of a “super bulky” ligand, designed to support metals in unusually low coordination geometries. We have successfully metalated this “super bulky” ligand with iron and are now exploring its metalation chemistry with other 3d metals. Recently, treatment of “super-bulky ” with CoCl2 afforded a red-‐orange solid (Scheme 1) that we believe contains Co.
Project Description: We recently reported the synthesis of a “super bulky” ligand, designed to support metals in unusually low coordination geometries. We have successfully metalated this “super bulky” ligand with iron and are now exploring its metalation chemistry with other 3d metals. Recently, treatment of “super-bulky ” with CoCl2 afforded a red-‐orange solid (Scheme 1) that we believe contains Co.
If successful, this would open new chemical avenues to explore.
Need for single crystal x‐ray diffraction method:
As our complex is paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization isambiguous for determining thecompositionand connectivity/structure ofthe complexes. Other characterization techniques like infrared (IR), ultraviolet-‐visible (UV-‐Vis), electron paramagnetic resonance (EPR) are also gave us inconclusive data for the structure/connectivity of the complexes. For such paramagnetic complexes single crystal diffraction study is found to be most successful. The crystal structure determination will conclusively tell us aboutthe identity of this product.
*This compound isair and water sensitive and will require collection under an N2 cryostream.
LiquidN2 can be provided upon request.
Solvents/chemicals used: Tetrahydrofuran (THF), diethyl ether (Et2O),and hexanes.
Unsupported Metal-Metal Bonds Involving Uranium
PI:Dr. Skye Fortier (Chemistry)
 Project Description: Metal-metal bonded complexes have played and important and critical role in the development of modern, synthetic inorganic chemistry. Moreover, these complexes have provided invaluable insight into the electronics and bonding modes of the transition metals. In contrast, M-M bonded complexes containing an actinide metal are rare and poorly studied. We have developed a new ligand scaffold (abbr. Lar) that we believe can provide ancillary support for the stabilization of U-M bonds. We recently performed a reaction between LarUI with an anionic metal synthon (CpFe(CO)2-) (Scheme 1). While a reaction was observed, spectroscopic data does not indicate whether a new U-Fe compound has been successfully formed.
Project Description: Metal-metal bonded complexes have played and important and critical role in the development of modern, synthetic inorganic chemistry. Moreover, these complexes have provided invaluable insight into the electronics and bonding modes of the transition metals. In contrast, M-M bonded complexes containing an actinide metal are rare and poorly studied. We have developed a new ligand scaffold (abbr. Lar) that we believe can provide ancillary support for the stabilization of U-M bonds. We recently performed a reaction between LarUI with an anionic metal synthon (CpFe(CO)2-) (Scheme 1). While a reaction was observed, spectroscopic data does not indicate whether a new U-Fe compound has been successfully formed.
Need for single crystal x‐ray diffraction method:
As our complex is paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization is ambiguous for determining the composition and connectivity/structure of the complexes. Other characterization techniques like infrared (IR), ultraviolet-visible (UV-Vis), electron paramagnetic resonance (EPR) are also gave us inconclusive data for the structure/connectivity of the complexes. For such paramagnetic complexes single crystal diffraction study is found to be most successful. The crystal structure determination will conclusively tell us about the identity of this product.
*This compound is highly air and water sensitive and will require collection under an N2 cryostream. Liquid N2 can be provided upon request.
Solvents/chemicals used: Dimethoxyethane (DME), diethyl ether (Et2O), and hexanes.
H-Atom Reactivity of a Super Bulky Aniline
PI:Dr. Skye Fortier (Chemistry)
 Project Description: We recently reported the synthesis of a “super bulky” ligand, designed to support metals in unusually low coordination geometries. This ligand is synthesized from a “super bulky” aniline precursor. This precursor, see scheme below, possesses benzylic H-atoms which are known to be reactive functionalities and susceptible to H-atom abstraction. We have observed reactivity at this position and have undertaken a study to better study this chemistry at a fundamental level. Oxidation of the “super bulky” aniline with PbO2 in dimethoxyethane gives a deep red solution from which orange crystals can be isolated in very low yields.
Project Description: We recently reported the synthesis of a “super bulky” ligand, designed to support metals in unusually low coordination geometries. This ligand is synthesized from a “super bulky” aniline precursor. This precursor, see scheme below, possesses benzylic H-atoms which are known to be reactive functionalities and susceptible to H-atom abstraction. We have observed reactivity at this position and have undertaken a study to better study this chemistry at a fundamental level. Oxidation of the “super bulky” aniline with PbO2 in dimethoxyethane gives a deep red solution from which orange crystals can be isolated in very low yields.
Need for single crystal x‐ray diffraction method:
As our complex is paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization is ambiguous for determining the composition and connectivity/structure of the complexes. Other characterization techniques like infrared (IR), ultraviolet-visible (UV-Vis), electron paramagnetic resonance (EPR) are also gave us inconclusive data for the structure/connectivity of the complexes. For such paramagnetic complexes single crystal diffraction study is found to be most successful. The crystal structure determination will conclusively tell us about the identity of this product.
*This compound is air and water sensitive and will require collection under an N2 cryostream. Liquid N2 can be provided upon request.
Solvents/chemicals used: Dimethoxyethane (DME) and hexanes.
A W2 complex with a bond order of five
PI:Dr. Dino Villagran (Chemistry)
Students: Karen Ventura
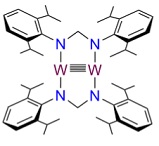 Project Description: Fifty years ago, the discovery of the first quadruply bonded complex, [Re2Cl8]2–, initiated a paradigm shift the understanding of inter-metal bonds and shook up the field of inorganic chemistry. Since then, hundreds of bimetallic complexes with different bond orders and electronic properties, mostly involving group 6 metals, have been synthesized and structurally characterized. The next step in inter-metal multiple bonds was made in 2005 when the first stable dichromium molecule having a bond order of five was reported, followed shortly by analogous Mo molecule. In this study we present the synthesis and characterization of a novel ditungsten complex with a bond order of five. We are uniquely interested in the whether this complex contains D2h core symmetry defined by two N,N′-bis(2,6-diisopropylphenyl)formamidine ligands are positioned in a trans fashion. Single crystal X-ray will allow us to demonstrate the fine structure and symmetry.
Project Description: Fifty years ago, the discovery of the first quadruply bonded complex, [Re2Cl8]2–, initiated a paradigm shift the understanding of inter-metal bonds and shook up the field of inorganic chemistry. Since then, hundreds of bimetallic complexes with different bond orders and electronic properties, mostly involving group 6 metals, have been synthesized and structurally characterized. The next step in inter-metal multiple bonds was made in 2005 when the first stable dichromium molecule having a bond order of five was reported, followed shortly by analogous Mo molecule. In this study we present the synthesis and characterization of a novel ditungsten complex with a bond order of five. We are uniquely interested in the whether this complex contains D2h core symmetry defined by two N,N′-bis(2,6-diisopropylphenyl)formamidine ligands are positioned in a trans fashion. Single crystal X-ray will allow us to demonstrate the fine structure and symmetry.
Need for single crystal x‐ray diffraction method: Fine structure data will assist greatly in determining the core symmetry. Other methods can provide composition, but only diffraction techniques can provide this geometric information. The product is air and water sensitive. The nitrogen cryostream (Oxford) equipped diffractometer (Bruker) is required to keep the sample in an inert environment for data collection.
Novel functional 2D crystals for nano-device application
PI:Dr. Anupama B. Kaul
Students/Staff:Dr. Dalal Fadil (Postdoctoral Fellow)

Project Description: In contast to graphene, 2D transition metal dichalcogenides (TMD) such as molybdenum disulfide (MoS2) exhibit an attractive band gap from 1.29 eV in the bulk crystal to 1.90 eV for monolayers. Moreover, there is an indirect to direct band gap transition in MoS2 as it is thinned to monolayers from the bulk. Given the pristine interface of 2D materials with the absence of dangling bonds, transport and scattering are confined to the plane of the material. The structure of layered MoS2 is shown in the figure where Mo atoms are sandwiched between sulfur atoms on either side and the covalent interaction is responsible for the strong intralayer bonding. In contrast the interlayer bonding is through the weak van der Waals interaction.
Need for single crystal x‐ray diffraction method: We would like to study the structural changes that occur in powdered MoS2 samples as you heat them both in air and in an inert vacuum environment. MoS2 appears to have issues with stability in an oxidizing environment at elevated temperatures. Through this XRD analysis conducted in the various environments, we would like to gain insights into the structural transformations that may be occurring in this material which will impact both its properties and the applications that can emerge from this material.
Transition metal sulfides – catalysts for hydrogenation and hydrogenolysis reactions
PI: Professor Russell R.Chianelli
Student/StaffI: Juan Leal
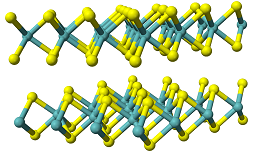
Project Description: The transition metal sulfides (TMS) are a class of catalysts that are able to facilitate numerous hydrogenation and hydrogenolysis reactions while in the presence of sulfur. These reactions are integral to the refinement process currently used in the petrochemical industry. The catalytic activity and selectivity of the TMS stems from the electronic and structural properties of the sulfides. The d electrons in the frontier orbitals of the catalysts are key in directing catalysis at the surface. The d electrons available are a function of the transition metal selected and, where applicable, a transition metal containing material used as a promoter. Recently, promising results have been obtained with molybdenum sulfide and a cobalt containing promoter material, however the synthesis reaction conditions still require some tuning in order to optimize the catalytic potential of our system. Commercialization of the catalytic materials produced are in the process of commercialization with POROCEL Int.
Need for x-ray methods / use of XRSLab:
Samples of the catalyst were taken after being subjected to a range of reaction conditions for predetermined lengths of time. Powder XRD is required to measure the quality of the sample and quantify any degradation which can occur.
Silver N-Oxide Isonicotinic Acid Crystal Channels for Desalianation
PI: Dr. Juan Noveron (Chemistry)
Student/StaffI: Jacobo Garcia

Project Description: Metal-organic coordination polymers (MOCP) have been found to exist in a variety of three dimensional structures with tunable chemical and physical properties. Cavities and channels of various shapes and sizes can be incorporated into these organic-inorganic crystalline structures by choosing appropriate reaction conditions and materials. Among these structures, there have been a number of stable structures characterized by single crystal X-ray diffraction in which the initial supramolecular framework encapsulates water clusters in channels. Further, these architectures possess crystal memory that allow for dehydration-rehydration properties, shown to be reversible. The scope of these findings makes the properties of a synthesized silver N-oxide complex framework a potential application in the desalination of water. X-ray powder diffraction will confirm the synthesis of these crystals which will streamline the synthesis portion of the project. In this project, the aim is to develop a filter material in which the channels created in the network structure of silver N-Oxide isonicotinic acid can be used to desalinate water as it passes through the framework.
Need for x-ray methods / use of XRSLab:
High-resolution powder diffraction is a needed to verify the correct structure of the complex obtained in the experimental procedure. This technique is necessary to move forward with this project and start vital experimentation for the application of the material as a water filter.
Functionalization of CeLu2N@Ih-C80
PI: Dr. Luis Echegoyen
Student/StaffI: Danisha M. Rivera-Nazario, Catalina Suarez, Paulina A. Quiñonez

Project Description: Several endohedral fullerenes of the type M3N@C2n have been synthesized and isolated, and these are the most studied endohedral cluster fullerene types because of their relatively easy preparation and the vast number of possible trimetallic nitride clusters that have been incarcerated to date. However, adding a different metal atom to the incarcerated cluster increases the complexity of the purification and functionalization, because the nature of the encapsulated cluster influences the exohedral functionalization behavior of endohedral fullerenes.To the best of our knowledge there is only one report of the functionalization of mixed metallic nitride EMF but no information is available of how the mixed metal cluster affects the surface chemistry of the cage.We thus decided to perform a 1,3-dipolar cycloaddition reaction with N-ethylglycine and its reactivity and electrochemical properties were investigated.
Need for x-ray methods / use of XRSLab:
High-resolution Powder XRD would allow the characterization of the complex fullerene structures which aids in the study of functionalization.
Making of Three Coordinate Iron
PI: Dr. Skye Fortier
Student/StaffI:Arnab K Maity (Postdoc)

Project Description: We are very interested in low coordinate complexes, especially ‘three-coordinate complexes’, in which the metal has only three bonds. Typically, a metal possesses six and four bonds in an octahedral and tetrahedral geometry, respectively. These three coordinate complexes, which are very rare, can be studied for new chemistry. For example, low-coordinate complexes are electronically unsaturated, whereas most complexes prefer an 18-electron configuration. The low coordinate complexes have only 12-14 valence electrons, thus, leading to unusual reactions. Additionally, the third ligand may be varied for electronic and steric control of the reactions. In particular, we are trying to synthesize such complexes using a ‘super-bulky’ guanidinate platform on iron. We have recently published the synthesis and the characterization of the ‘super bulky’ guanidinate, and are now in pursuit of its chemistry on iron. In this context, we are in the process of generating ‘three coordinate iron’. One such reaction is between ‘super bulky’ guanidinate Fe-ClTHF and [(NtBu)Imidazoline-2-ylide guanidinate Fe(tBu-NHC)]B(C6F5)4] - our hope is that this will lead to three coordinate [‘super bulky’ guanidinate Fe(tBu-NHC)]B(C6F5)4].
Need for x-ray methods / use of XRSLab:
As our 'super bulky iron' species are predominantly paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization is not ambiguous for knowing the composition and the connectivity/structure of the complexes. Other characterization techniques, such as, infrared (IR) and ultraviolet-visible for such paramagnetic 'iron' complexes single crystal diffraction study found to be most successful. In this context, we have grown some single crystals of the products from a 'super bulky iron chloride + tBuNHC +K[B(C6F5)4]'reaction suitable for X-ray diffracttion. The crystal structure determination will inconclusively tell us about the identity of these products. These compounds are highly air and moisture sensitive and will require collection under N2 cryostream. Liquid N2 can be provided upon request. Solvents/chemical used: Toluene, Et20,Pentane.
Reductive Elimination of Palladium complex
PI: Dr. Dino Villagran (Chemistry)
Student/StaffI: Karen Ventura and YanyuWu

Project Description: Reductive elimination is one of the most fundamental transformations in redox catalysis of organometallic chemistry, which describes the process of the oxidation state and the coordination number of the metal decrease by 2 and with liberation of a small molecule. Although reductive elimination of mononuclear complex has been studied massively, the mechanisms of redox chemistry of multinuclear complex are less understood, while redox chemistry of biological catalysis often happens at multinuclear sites.Palladium complexes have been playing an important role in catalysis of a lot of chemical reactions. We wish to study a bi-palladium complex supportedby a formamidineligand for the examinationof reductive elimination. As well as a bi-molybdenum complexof a higher bond order supported by aguanidinateligand system. The characterization and detailed information about these metal environments are key to understanding the factors to the desired pathway of reductive elimination.
Need for x-ray methods / use of XRSLab:
Fine structure data will assist greatly in determining the core symmetry. Other methods can provide composition, but only diffraction techniques can provide this geometric information. The product is air and water sensitive. The nitrogen cryostream (Oxford) equipped diffractometer (Bruker) is required to keep the sample in an inert environment for data collection.
Trapping of 'Iron Nitrido' by External Nucleophiles
PI: Dr. Skye Fortier
Student/StaffI:Arnab K Maity (Postdoc)

Project Description: Highvalent iron(IV/V) nitrido species have been spectroscopically identified as active intermediates in the catalytic cycles of dinitrogen fixation by enzyme Nitrogenase. Also, these species are believed to be active intermediates in the industrial dinitrogen fixation in Haber Bosch process. The synthesis of well-described model compounds can provide vital insights in to the mechanism of such processes. We, in particular, are trying to develop such models using a ‘super-bulky’ guanidinate platform on iron. We have already published the synthesis and characterization of the ‘super bulky’ guanidinate recently and now in pursuit of its chemistry on iron. In this context, we are in a process to generate ‘iron-nitrido’ which seems to be highly reactive and undergoes intramolecular quenching. To better understand the geometry and reactivity of this putative ‘ironnitrido’ we are using external nucleophile to trap such intermediate species. One such reaction, in particular with trimethyl phosphine, gave us some promising results and if we are successful we will be able to study more about such rare ‘iron-nitrido’ species.
Need for x-ray methods / use of XRSLab:
As predominantly our ‘super bulky iron’ species are paramagnetic in nature, any nuclear magnetic resonance (NMR) based characterization is not ambiguous for knowing the composition and connectivity/structure of the complexes. Other characterization techniques like infrared (IR), ultraviolet-visible (UV-Vis), electron paramagnetic resonance (EPR) are also gave us inconclusive data for the structure/connectivity of the complexes. For such paramagnetic ‘iron’ complexes single crystal diffraction study is found to be most successful. In this context, we have grown some single crystals of the products from a ‘super bulky iron azide photolysis + PMe3’ reaction suitable for X-ray diffraction. The crystal structure determination will conclusively tell us about the identity of these products.
Possible Structures: *These compounds are highly air and water sensitive and will require collection under an N2 cryostream. Liquid N2 can be provided upon request.
Solvents/chemicals used: Diethyl ether, Toluene, Hexane, pyridine
CeO2 Nanoparticles
PI: Dr. Jorge Gardea-Torresdey (Chemistry)
Student/StaffI:Ana Barrios and Mena Zuverza

Project Description: Iron and copper nanomaterials are widely used in environmental remediation and agriculture. However very little is known about the effects of core-shell CeO2 NP’s on macro and micronutrient uptake and stress enzyme activities on edible plants. This study requires nanoparticle characterization of CeO2 NP’s through comprehensive X-Ray Diffraction and subsequent Scherrer calculations to determine nanoparticle size in comparison of un-coated and citric acid coated nanoparticles.
Need for x-ray methods / use of XRSLab:High-resolution powder diffraction is an adequate tool for the above-mentioned structural studies because of its ability to reveal subtle crystallographic modifications with sub-Ångstrom resolution. Using the variable peak width as a result of nanoparticle peak broadening phenomena which relates the size of sub-micron particles to the broadening of peaks in a diffraction pattern. The ability to use High Score Plus© software will allow us to quickly and accurately calculate nanoparticle size and surface strain.
Synthesis toward linear fullerene polymer and 2D layer
PI: Dr. Luis Echegoyen (Chemistry Department)
Student/StaffI:Dr. Shuming Liu (Postdoctoral researcher)

Project Description: Metal–organic frameworks (MOFs) are a relatively new class of hybrid organic-inorganic polymers derived from ordered networks formed from organic ligands and secondary building units or metal ions. Since MOFs exhibit permanent porosity, they have attracted considerable interest and have been extensively studied because of their structural diversity, intrinsic variety of topologies, and unusual properties. These new hybrid materials are promising for applications in nonlinear optics, gas storage, catalysis, and for chemical separations. The synthesis of new MOFs is very challenging, with many choices of metals and organic ligands. Different combinations of metal centers and organic ligands have resulted in elaborate designs with interesting structures and properties.
Starting from fullerene derivative compound 1 which has four malonates and two diazafluorene groups on trans-1 positions of the fullerene, we synthesized a series of compounds with similar structure to compound 2. Under different conditions, compound 2 could be a bimetallic complex, zig-zag linear polymer or linear polymer.
Need for x-ray methods / use of XRSLab: Future work will connect different lines of compound 2 with metals to coordinate with the malonates. By controlling the ratio of metal to compound 2, 2D or 3D MOFs are expected. X-ray diffraction will aid in the determination of the structure of compound 2.
Influence of graphite sources and oxidation methods on the conductivity of Reduced Graphene Oxide Paper
PI: Dr. Russell Chianelli
Student/StaffI:Eva Deemer (MASE Ph. D. Interdisciplinary program)
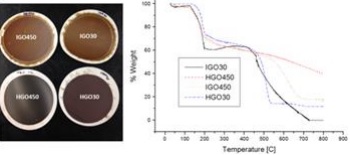
Project Description: Graphene is a promising material that has extraordinary mechanical, electronic , magnetic and optical properties. However, there currently remains a high market barrier for large scale graphene producers due to lack of material standards. This challenge for producers to describe graphene quality with respect to lateral dimension and exfoliated layers has enormous consequence on the application and commercialization of graphene. Understanding the influence of oxidation and parent graphite have on synthetic chemical methods is important for functionalization reactions and for producing high quality graphene. The results presented can aid in the development of materials used in transparent conductive layers for solar cells, transparent electrodes, coatings, sensors and other nano-electronic devices. This work can be useful for material scientists or aid synthetic chemists that are interested in the role graphite sources and oxidation path have on determining the quality of resulting reduced graphene.
Need for x-ray methods / use of XRSLab: Powder diffraction and grazing angle incidence XRD (GIXRD) is useful for studying the structure of resulting oxidized graphene papers by identifying interlayer spacing. It is difficult to resolve since the structural difference are identified between 0º-10ºϴ.




